PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
Decomposes
REFRACTIVE INDEX
NFPA RATINGS
AUTOIGNITION
199 C
FLASH POINT
APPLICATIONS
Camphor is a white crystalline solid cyclic ketone with a musty aroma and a characteristic taste. It is slightly cold to the touch like menthol. It is practically insoluble in water but soluble in alcohol, and other solvents icluding ether, chloroform. It melts at 176°C and boils at 204°C. It obtained from the Asian tree Cinnamomum camphora. It is isolated by passing steam through the pulverized wood and condensing the vapours; camphor crystallizes from the oily portion of the distillate and is purified by pressing and sublimation. It is also manufactured synthetically by several processes from the compound a-pinene obtained from oil of turpentine. Camphor is widely used as a plasticizer in the manufacture of celluloid and some lacquers. It is used in medicine internally for its calming influence as a stimulant, a diaphoretic, and an inhalant and externally as a counter-irritant in rheumatisms, sprains bronchitis , and in inflammatory conditions. Camphor is also an ingredient in cough remedies, ear drops, and preparations for the removal of corns and verrucas.
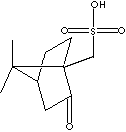
Sulfonic acid is a compound with general formula RSO2OH, where R is an aliphatic or aromatic hydrocarbon. It is a derivative of sulfuric acid (HOSO2OH) where an OH has been replaced by a carbon group or a compound where a hydrogen atom has been replaced by treatment with sulfuric acid; for example, benzene is converted to benzenesulfonic acid (water-soluble). Sulfonic acid has a sulfur atom bonded to a carbon atom of a hydrocarbon and bonded also to three oxygen atoms, one of which has been attached to a hydrogen atom. Sulfonic acid is acidic due to the hydrogen atom, stronger than a carboxylic acid. Sulfonic acid is one of the most important organo sulfur compounds in organic synthesis. Sulfonic acids are used as catalysts in esterification, alkylation and condensation reactions. Sulfonates are salts or esters of sulfonic acid. Sulfonic salts are soluble in water. Sulfonic acid and its salts present in organic dyes provide useful function of water solubility and or improve the washfastness of dyes due to their capabiltity of binding more tightly to the fabric. They are widely used in the detergent industry. Alkylbenzene sulfonic acid is the largest-volume synthetic surfactant because of its relatively low cost, good performance, the fact that it can be dried to a stable powder and the biodegradable environmental friendliness. Sulfonate cleaners do not form an insoluble precipitates in hard water. Sulfonic acid salts and esters are intermediates widely used in organic synthesis and particularly phenolic compounds and cation exchange resins. They are synthetic intermediate for a number of biologically active compounds and pharmaceutical candidates such as sulfa drugs.APPEARANCE
ASSAY
98.0 - 102.0%
MELTING POINT
SPECIFIC ROTATION
2.0% max
HEAY METALS
20ppm max
0.005% max